Extreme radiation resistance achieved by mutations in DNA repair mechanisms of black yeasts.1/5/2021 Reporting on: Romsdahl J, Schultzhaus Z, Chen A, Liu J, Ewing A, Hervey J, Wang Z. 2020. Adaptive evolution of a melanized fungus reveals robust augmentation of radiation resistance by abrogating non‐homologous end‐joining. Environmental Microbiology. [link]  Ramsar, Iran. Source: Wikimedia Commons. Ramsar, Iran. Source: Wikimedia Commons. It goes without saying that ionizing radiation can be a scary thing. This “invisible force" can dismantle you from within by breaking your DNA to pieces. You’d need a hefty dose to suffer such consequences, though. In fact, we’re exposed to relatively harmless levels of background radiation regularly. For example, residents of Ramsar, Iran—the city that holds the current record for highest background radiation—are exposed to an average dose of 10 Gy per year [1]. (The Gray (Gy) is a dose unit that reflects the amount of energy delivered to a mass of tissue.) To give you some perspective, in the aftermath of the 1986 Chernobyl disaster, evacuees were reported to have received a whole-body dose of 0.002 to 0.66 Gy [2]. Ramsar's natural ionizing radiation comes from its geology, which is dotted by hot springs that move radioactive decay to the earth’s surface. Surprisingly, there’s been no increase in rates of cancer, leukemia, or other radiation-associated health complications among the residents [1]. Some researchers even argue that low to moderate exposure to ionizing radiation can have a therapeutic effect by activating DNA repair systems and other protective mechanisms in our bodies—not far off from how vaccines work [3]. However, after a certain threshold, there’s no escaping the damage that ionization radiation can inflict. The average acute dose that would cause radiation sickness is 0.7-10 Gy [4]. No human is expected to survive an exposure higher than 10 Gy, and at 50 Gy, death is expected within three days [4]. Keep in mind that these are acute thresholds—the same doses spread over a longer time may not be so damaging because the body will have time to repair between exposures. Unlike non-ionizing radiation (e.g., microwaves and UVA light), which only has enough energy to produce heat, ionizing radiation can break atomic bonds (i.e., damage to the body at the molecular level) and it’s penetrative, so damage can be inflicted internally. There are four types of ionizing radiation: alpha (α), beta (β), neutrons, and electromagnetic waves such as gamma (γ) rays. Alpha radiation particles are the heaviest and are emitted by naturally occurring radioactive materials like uranium and radon (e.g., in smoke detectors). Because they’re so heavy, they can’t penetrate barriers very well-- they can’t even get through paper (although inhalation can pose problems). Beta particles consist of single electrons and are emitted by radioactive isotopes of elements like hydrogen (e.g., tritium) and carbon (e.g., carbon-14). Beta particles don’t penetrate very well either and can be stopped by a layer of clothing. Neutrons are a form of ionizing radiation that consist of, well, free neutrons. They’re commonly produced from the splitting of atoms in nuclear reactors. Neutrons can travel a long way and can penetrate most barriers, even lead. The only way to stop them is with large quantities of water or other materials made of very light atoms, such as boron, which can bounce and absorb them (concrete is often used in this capacity). Finally, we have electromagnetic waves, like X-rays and γ rays, which are commonly used in medicine because they can penetrate our bodies. They’re produced when a particle, like an electron, is accelerated by an electric field or by the radioactive decay of atomic nuclei. 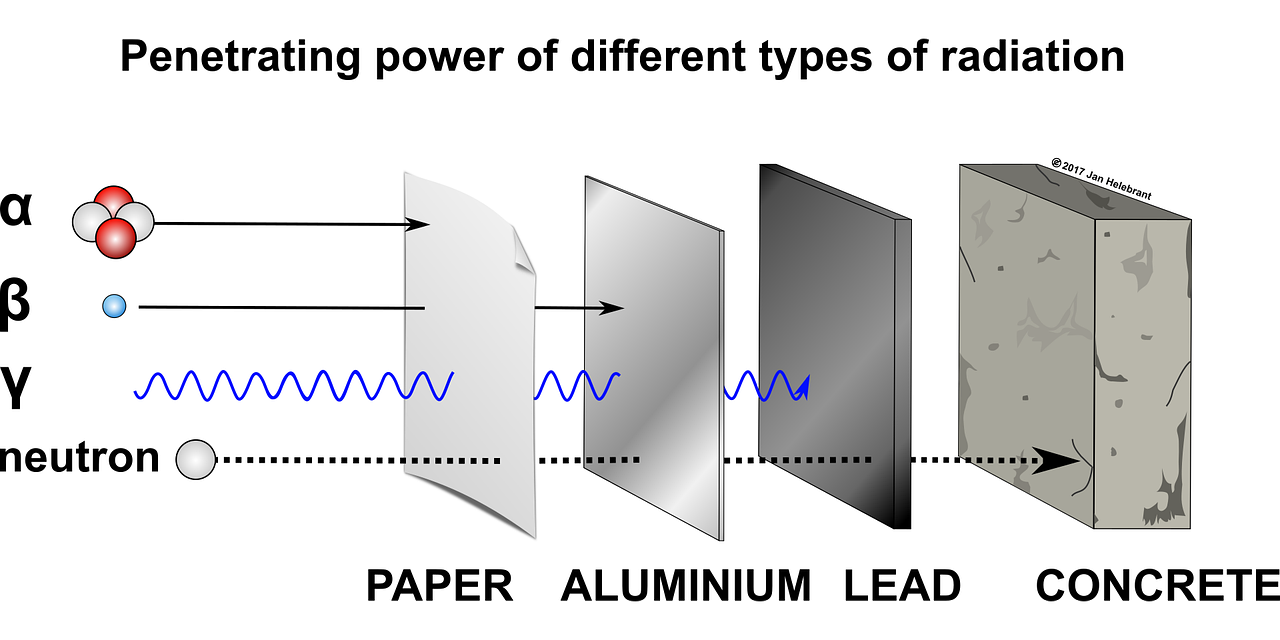 Penetration capacities of ionizing radiation. Source: Image by Jan Helebrant from Pixabay.  Species of black yeast known as Exophiala phaeomuriformis. Source: Wikimedia Commons. Species of black yeast known as Exophiala phaeomuriformis. Source: Wikimedia Commons. Now that we have a basic understanding of ionizing radiation, let’s talk about… yeast. Fungi are among the most radiation resistant eukaryotic organisms on Earth. Black yeasts in particular have been found in highly radioactive environments, such as the cooling pools of nuclear reactors, the stratosphere, and the damaged nuclear reactor at Chernobyl [5]. The remarkable resistance of black yeast to ionizing radiation presents an opportunity for scientists to pinpoint the genetic and biochemical underpinnings of this adaptation and consequently inform efforts aimed at developing therapies and safeguards against ionizing radiation. This can be of particular use to solving a major limitation of human space exploration: long-term exposure to cosmic ionizing radiation imposed by the space environment [6]. Several studies have attempted to uncover the genetic underpinnings of radiation resistance by searching for adaptive mutations in the genomes (i.e., the full DNA content) of resistant organisms [7]. This basically involves comparing the genetic code of a highly resistant species to that of a closely related non-resistant species, looking for differences (e.g., mutations), then investigating those differences. For example, do they occur in genes that code for enzymes involved in DNA repair? Unfortunately, these studies have been more or less inconclusive due to the sheer expanse of genomic data and the complexity of how mutations facilitate novel abilities. Taking this a step further, some researchers have attempted to artificially produce radiation resistance in bacteria. Essentially, this involves blasting bacteria with radiation, selecting and growing the survivors, then repeating this over many generations until a highly resistant strain emerges. This method is known as "directed laboratory evolution" and through such experiments, scientists have repeatedly found that changes in genes coding for DNA repair proteins facilitated the evolution of radiation resistance [8, 9, 10]. In other words, bacteria "artificially" evolved resistance to ionizing radiation by improving their DNA repair mechanisms. In October 2020, Drs. Jillian Romsdahl and Zheng Wang of the Naval Research Laboratory in Washington, DC, and colleagues published the results of a directed evolution study involving black yeast, which revealed a surprising adaptive mechanism against radiation. After repeated rounds of artificial selection, Romsdahl et al. produced several strains of extremely radiation resistant black yeast. Following genetic examination and functional experimentation, they discovered that these highly resistant strains owed a portion of their resistance to the deactivation of one of their DNA repair mechanisms. That may seem completely counterintuitive, but hear me out because this is really cool… Here’s how they did it: black yeast, which already have resistance to ionizing radiation were subjected to extremely high doses of γ radiation. The survivors were grown and then subjected to another round of γ radiation. This process was repeated 15 times, and the amount of radiation they were subjected to was increased every five rounds. In the first five rounds, they were given a 4500 Gy dose of ionizing radiation (remember the lethal dose for a human is anything higher than 10 Gy!). In the next five rounds the survivors received 5000 Gy, and in the final five, 5500 Gy. The average survival rate in each round was 1%. The yeast were essentially directed to evolve even more resistance than they already had. Since DNA damage is accepted as the main cause of death from ionizing radiation, Romsdahl et al. proceeded to subject their newly evolved strains with all sorts of DNA damaging methods, including exposure to desiccation, UVC light, the carcinogen methyl methanesulfonate, and anti-cancer drugs like Bleomycin and Hydroxyurea. They found that although their newly evolved strains had acquired higher resistance to ionizing radiation, for several strains this came at a cost to their ability to repair other types of DNA damage and to their general fitness. Next, Romsdahl et al. sequenced and analyzed the full genomes of all the evolved strains to identify possible causative mutations—they found many. By examining the functional significance of these mutations, one clear pattern emerged: the repeated occurrence of detrimental mutations (i.e., frame shifts or premature stop codon gains) in genes involved in a DNA repair method known as “non-homologous end joining” (NHEJ), basically rendering the mechanism nonfunctional. This is surprising considering that NHEJ repairs DNA damage and you’d think you'd want that up and running when you’re blasted with ionizing radiation. Well, not exactly… To investigate this puzzling outcome further, Romsdahl et al. took fresh black yeast strains that had been genetically modified to not have certain genes associated with NHEJ (i.e., they didn’t have functioning NHEJ), and exposed them to 4000 and 6000 Gy of γ-radiation. Lo and behold, they found that the deletion of these genes resulted in increased resistance to γ-radiation. However, the most resistant of the evolved strains still demonstrated higher resistance than the non-evolved, genetically modified strains. This means that the genetic underpinnings contributing to the evolved strains’ resistance are more complex and likely involves several altered mechanisms, beyond just the deactivation of NHEJ. 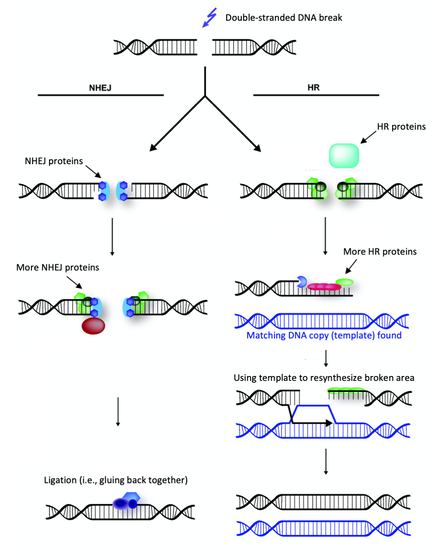 NHEJ vs HR. Source: modified from Wikimedia Commons. NHEJ vs HR. Source: modified from Wikimedia Commons. So, what is NHEJ and how could its deactivation help against ionizing radiation? NHEJ goes hand in hand with another DNA repair mechanism know as homologous recombination (HR). Both function in repairing double stranded DNA breaks—something that ionizing radiation is very good at inflicting. NHEJ fixes a break by directly ligating (i.e. gluing) the broken ends back together. HR repairs breaks by using a gene template (we have two copies of each gene—HR finds the copy and uses it as a template), which serves as a validating step to make sure the repair is accurate—something NHEJ doesn’t have. Organisms with smaller genomes (i.e., less DNA), such as fungi and bacteria, typically have a stronger predisposition for HR because they don’t possess large regions of genetic code repeats that could potentially result in inaccurate repair through HR. Romsdahl et al. propose two hypotheses to explain why selection for radiation resistance would favor deactivation of the NHEJ repair system. The first is that it reduces error-prone repair. Repair of DNA breaks by NHEJ is generally a precise and highly efficient process. However, radiation-induced DNA breaks usually produce chemically altered, or “dirty”, DNA ends, which cause imprecise repair by NHEJ. Further, during extreme radiation exposure, you’ll likely get a lot of DNA breaks, and as a result increase the probability of NHEJ gluing the wrong ends back together—this would be lethal. The second hypothesis is that deactivation of NHEJ reduces competition for repair by HR proteins. In yeast, DNA break repairs are first tackled by NHEJ. Breaks that are left unrepaired by NHEJ are then handed over to HR. Given that NHEJ is recruited to the break site before HR, NHEJ proteins bound to the break site can delay HR initiation. Increasing HR efficiency by eliminating competition from NHEJ proteins substantially improves survival following exposure to a high ionization radiation dose. In fact, HR function has been reported to increase threefold when it doesn’t have to compete with NHEJ [11] You might now be wondering if this discovery is something that could be directly applicable to us. Could we increase our resistance to ionizing radiation by deleting a few of our NHEJ genes? After all, rapidly advancing genome editing technologies are continuously pushing us towards the realm of human gene modifications. Unfortunately, like all things in biology, it’s not that simple. Depending on the organism and specific situation, shunting from NHEJ to HR for repair may not be feasible because there could be substantial survival consequences for disrupting NHEJ. Such a switch would be more attainable for species that predominantly rely on HR, like yeast, than for those that more heavily favor NHEJ, like us. And based on my own work in genetic engineering, a mutation that produces a beneficial effect in the gene of one animal, can have a completely different effect in the same gene of a different animal—adaptive outcomes are highly context dependent. However, each new discovery, like this one by Romsdahl et al., contributes to our growing understanding of the complex evolutionary mechanisms by which new adaptations are facilitated. One day we’ll get to the applicable part. By Shab Mohammadi
0 Comments
Leave a Reply. |
AuthorWant to learn about the latest discoveries in science? Here, I provide exciting monthly news updates on topics related to evolution. Archives
May 2021
Categories |
Proudly powered by Weebly
 RSS Feed
RSS Feed