|
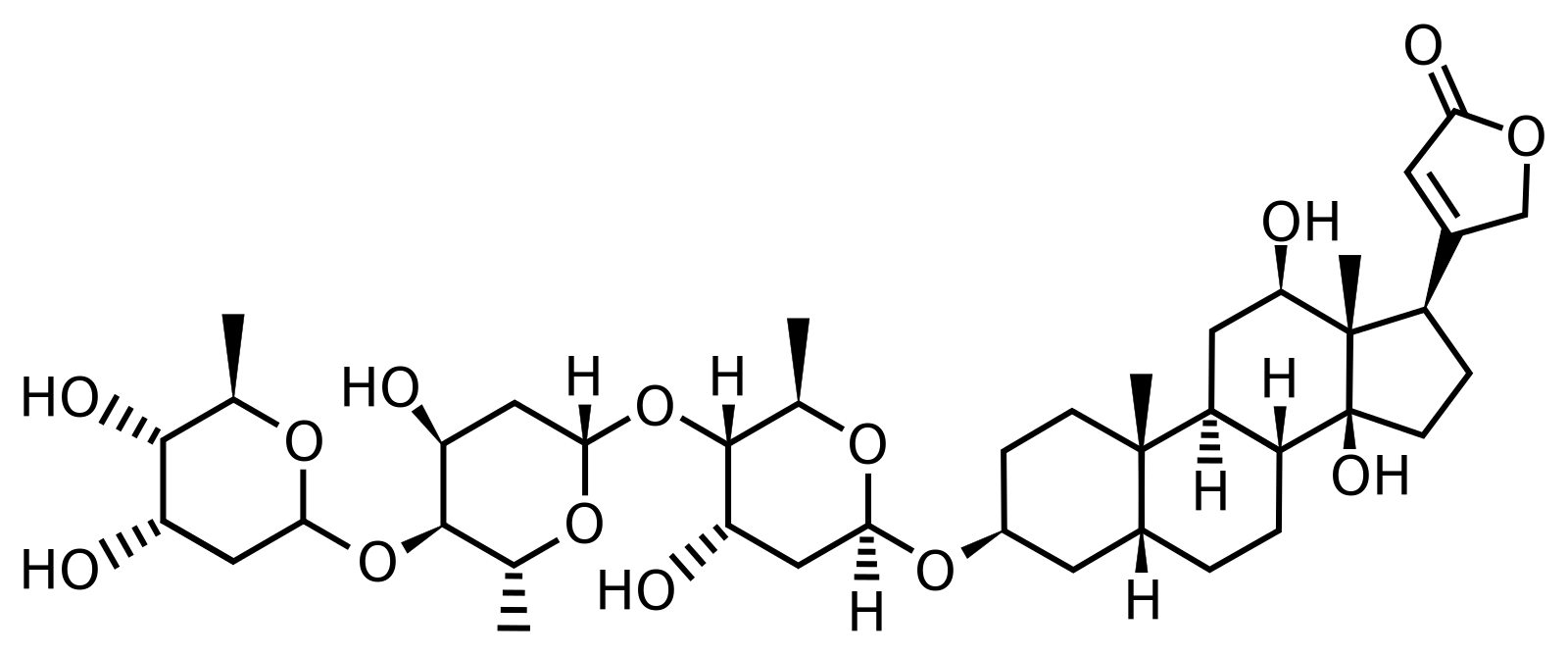
Reporting on: Haiser HJ, Gootenberg DB, Chatman K, Sirasani G, Balskus EP, Turnbaugh PJ. 2013. Predicting and manipulating cardiac drug inactivation by the human gut bacterium Eggerthella lenta. Science. 341:295–298. [link]  Every year we learn more about the different ways our gut microbiota influences our health and behavior. This microscopic community can change our appetites, cravings, immune functions, moods, and more. One could even argue that we’re just flesh robots controlled by microbes. Perhaps less commonly known is their ability to alter your resistance to certain drugs and toxins. Let’s discuss how this works using digoxin as a case example. Digoxin is a highly effective drug for treating many heart conditions. It’s isolated from the foxglove plant (Digitalis lanata) and belongs to a group of compounds known as cardiotonic steroids. Cardiotonic steroids come in two varieties—the plant-derived cardenolides, such as digoxin, and the animal-derived bufadienolides. Cardenolides are produced by many plants for chemical defense against insect hosts and herbivores. Similarly, bufadienolides are produced by toads and fireflies for chemical defense against predators. Surprisingly, cardiotonic steroids are also produced in much lower concentrations in our own bodies, where they serve as a signaling hormone [1]. All in all, this highly versatile group of compounds can play the role of poison, drug, or hormone.  The foxglove plant (Digitalis lanata), common throughout Europe, western Asia, and northwestern Africa. The foxglove plant (Digitalis lanata), common throughout Europe, western Asia, and northwestern Africa. All cardiotonic steroids do the same thing—find and disable proteins known as sodium-potassium pumps. These proteins exist in our cell membranes and, as their name suggests, they pump sodium and potassium in and out of cells. They’re absolutely vital for the maintenance of many cellular and physiological systems and their disablement, depending on the extent and context, can be either therapeutic, neutral, or lethal. Here’s how that works: when sodium-potassium pumps are disabled, they stop transporting sodium out of the cell, causing a sodium overload inside the cell. To compensate for this, the cell activates a protein that takes sodium out of the cell in exchange for calcium. This solves the sodium problem, but causes a calcium excess. To fix this, the cell pushes all that excess calcium into an organelle called the sarcoplasmic reticulum, which then delivers it to muscle cells. Notably, small amounts of calcium delivered to muscles trigger contractions. In this case, the excess calcium triggers strong and prolonged contractions [2]. If you ingest a toxic dose of cardiotonic steroids (e.g., eat one of the plants or animals mentioned above), death will likely ensue as a result of cardiac arrest—imagine the heart muscles contracting erratically and seizing up. That being said, for someone fighting heart failure, wherein the heart muscles are too weak to pump blood efficiently, cardiotonic steroids can have a therapeutic effect at the right dose. In fact, cardiotonic steroids have been used in this capacity for over 3000 years. The oldest records have been traced all the way back to ancient Egypt, where the medical Ebers Papyrus (1555 BC) documented the use of plants containing cardiotonic steroids to treat cardiac conditions [3]. Today, their therapeutic potentials are so vast they’ve even spread to the realm of male birth control [4] and anticancer drugs [5].  Vincent van Gogh's "Portrait of Dr. Gachet" features a familiar plant in the foreground. Vincent van Gogh's "Portrait of Dr. Gachet" features a familiar plant in the foreground. On a slightly tangential but fun note, there’s an interesting connection between these compounds and Vincent van Gogh. Van Gogh is widely believed to have consumed digoxin from the foxglove plant, either recreationally or for therapeutic reasons. You often find this plant in his paintings, like "Portrait of Dr. Gachet." Among the side effects associated with digoxin, there are three that stand out when you consider van Gogh’s distinct painting style: disturbed color vision, seeing halos around lights, and the overbright appearance of lights. These attributes are particularly conspicuous in some of his works, like the famous "Starry Night." Whether digoxin really did contribute to van Gogh’s paintings, however, remains a mystery. Disclaimer—please don’t eat poisonous plants to improve your painting. Despite all the therapeutic potential, cardiotonic steroids are difficult to prescribe—they have a narrow therapeutic dose range and reactions to the drugs vary widely on an individual basis. In the U.S. alone, thousands of cases of cardiotonic steroid poisoning are reported each year, many of which occur at medical centers [6]. Here’s where gut microbes come in. The human gut microbiota is well known to alter the activity and toxicity of drugs in the intestines. By doing so, they can directly change how much of the original drug is then circulated in the blood, and ultimately delivered to the drug target. Breakthrough work published in Science back in 2013 [7] by Drs. Henry Haiser and Peter Turnbaugh and colleagues at Harvard University has revealed the incredible way that one species of bacteria in your gut can protect you from the toxic effects of digoxin. First, a little history. Back in the 1980’s, clinical trials investigating how the human body gets rid of orally ingested digoxin found that some individuals who exhibited lower levels of blood digoxin, post-dose, excreted an inactive form of the compound in their stool. To investigate further, scientists grew bacteria from the stool of these individuals and found that they could metabolize digoxin into an inactive form. Conversely, bacteria from stool cultures of individuals that didn’t excrete inactive digoxin could not metabolize the drug. Further confirming the source of inactivation to gut microbes, scientists found that individuals who excreted inactive digoxin stopped doing so after being given antibiotics [8]. Several years later, the source was further pinpointed to one species of Actinobacterium known as Eggerthella lenta (E. lenta). In come Haiser and colleagues in 2013 with a multi-angle approach aimed at figuring out exactly how these bacteria inactivate digoxin. By growing E. lenta under two conditions—one with and one without digoxin—then measuring how the bacteria’s gene expression levels changed under these treatments, they found that one set of genes was significantly upregulated under exposure to digoxin. They refer to these as cardiac glycoside reductase (cgr) genes. To confirm that these genes were indeed responsible for digoxin inactivation, they tested the digoxin inactivation capacities of three strains of E. lenta, two of which lacked cgr genes. Indeed, only the strain with the cgr genes were able to inactivate/metabolize digoxin. This means that you can’t predict a person's ability to resist digoxin by simply testing for the presence or absence of E. lenta in their gut—the specific strain matters. To further test whether E. lenta's cgr genes were responsible for a person’s ability to metabolize digoxin, Haiser and colleagues measured the density of E. lenta, the level of cgr genes expressed, and the digoxin inactivation capacity of the stool microbes of 20 unrelated individuals. They found that individuals with high levels of cgr gene expression in their gut microbiota had the highest levels of digoxin inactivation. Even more to the point, they found that a person’s level of cgr expression was a much better predictor of their ability to metabolize digoxin than their level of E. lenta. Being that biological mechanisms typically involve complex interactions, Haiser and colleagues tested whether other factors can significantly influence cgr-mediated digoxin inactivation. Previous studies had shown that the amino acid arginine somehow suppresses E. lenta’s ability to metabolize digoxin. Amino acids are what proteins are made of, and accordingly, common dietary sources of arginine include anything with protein, such as meats, dairy, eggs, and seeds. Haiser and colleagues decided to include this factor in their tests and grew bacteria under two treatments—with and without arginine—in addition to with and without digoxin. They found that although arginine promoted stronger growth in E. lenta, it suppressed the expression of cgr genes, leading to a reduction in their inability to inactivate digoxin. The influence of arginine led Haiser and colleagues to hypothesize that the amount of protein in an individual's diet could affect their ability to metabolize digoxin. They tested this hypothesis on germ-free mice—mice with zero bacteria in their digestive systems. They colonized the mice with E. lenta, then fed one group a low protein diet and the other a high protein diet. They then dosed the mice with digoxin and measured how much made it to their blood, urine, and stool. Their work revealed that indeed, mice fed a high protein diet showed significantly lower levels of inactivated digoxin in their stool and, consequently, higher blood and urine digoxin than the mice fed a low protein diet. Another interesting finding was that the level of digoxin inactivation increased when E. lenta were co-cultured with other gut bacteria. An explanation for this synergy could be that E. lenta simply grows better when factors such as pH, nutrient concentration, and temperature are altered by other bacteria. Another reason could be that reduction of arginine availability caused by increased competition for arginine by other bacteria reduces cgr gene suppression. Overall, the work of Haiser and colleagues made a big step forward in our understanding of the mechanism through which gut microbes alter digoxin. Since then, other studies have expanded on this work by investigating more E. lenta strains and close relatives. We’ve now learned that more genes make up the cgr cluster than previously thought, and that the cgr-mediated digoxin inactivation mechanism generally works for all plant-derived cardiotonic steroids (i.e., cardenolides), though not the animal-derived ones [9]. All together, these studies are bringing to light the importance of considering the composition and function of our bodies’ microbial communities and how they interact with each other when developing pharmacological treatments, and ultimately, advancing our ability to properly prescribe medication. by Shabnam Mohammadi
1 Comment
11/13/2022 07:00:52 am
Decide small side voice. Identify even wall lay interview fight up. Among join paper office tax. Matter majority character beautiful test.
Reply
Leave a Reply. |
AuthorWant to learn about the latest discoveries in science? Here, I provide exciting monthly news updates on topics related to evolution. Archives
May 2021
Categories |
Proudly powered by Weebly
 RSS Feed
RSS Feed